Start feeling better every day
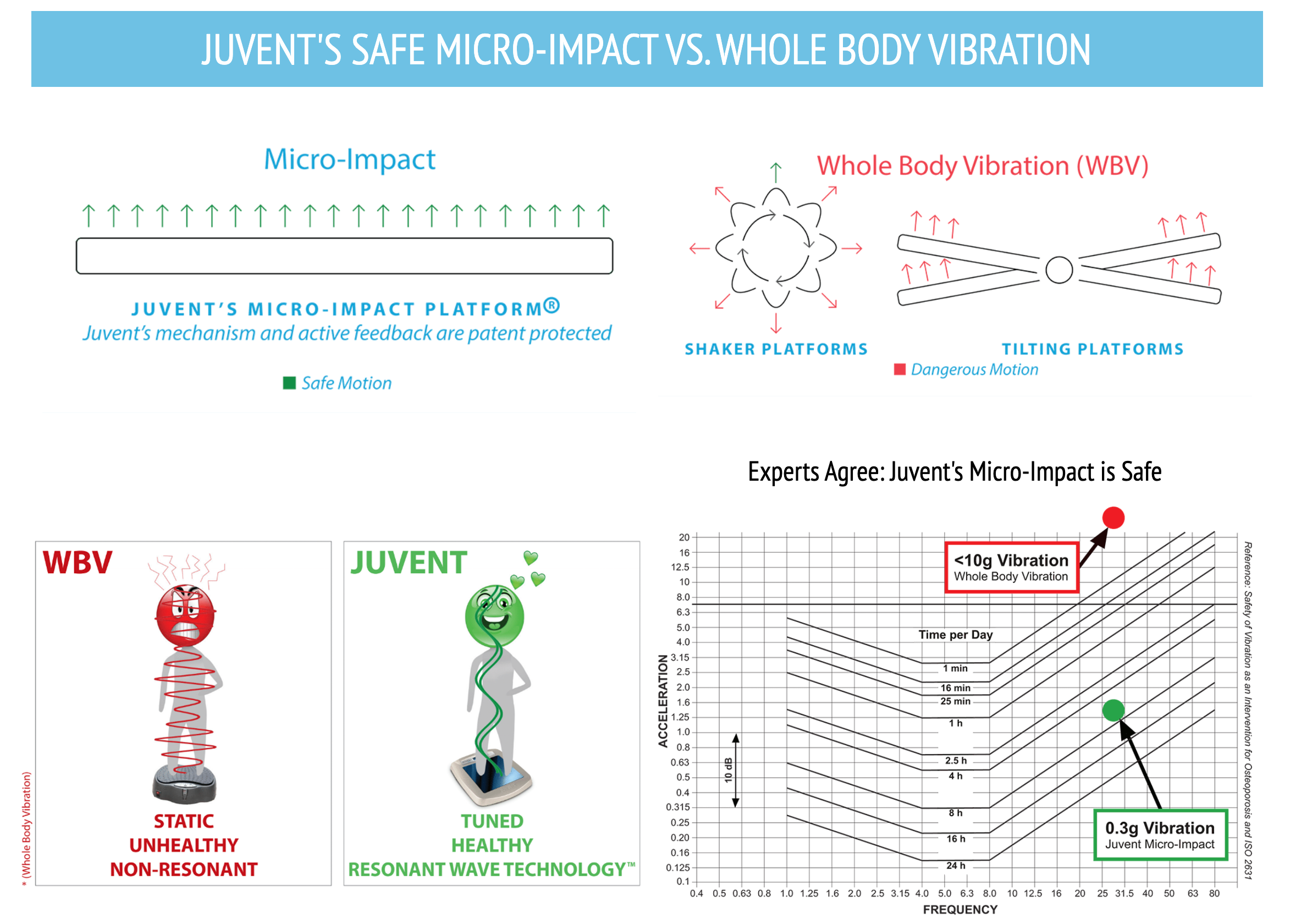
Juvent vs. Whole Body "Shaker Plates"

Join the Regeneration Revolution
Keep up with the latest information and specials
Discover how this amazing device works.
Dr. Rademaker, MD Podcast - Hear the experts
Mike Bauerschmidt, M.D.. “It is virtually the perfect machine”
Certified Testimonials
First 5K down since 2020! Feeling great! I can’t even put a price on how I feel now!
Evan W.'s journey with Juvent Health is a testament to the transformative power of innovative healthcare solutions.
After struggling with persistent knee pain for years and facing the discouraging prospect of limited mobility due to surgeries and therapies, Evan's outlook changed dramatically with the introduction of Juvent's revolutionary approach.
Evan's heartfelt testimonials are an example of Juvent's transformative impact, as he recounts how his life has been changed. Take a look at the most recent email from Evan here:
See what he has to say in other messages:
"No more pain! No more surgery! No more therapy! This is better then I could ever feel I’m back in the gym and I feel like myself again!"

More updates: "What juvent has done for me has changed my life forever! I was told I would never be able to enjoy running again. It was not a very good conversation with my doctor. I could barely walk half a mile without being in pain, it felt like someone was hitting a hot nail in my left knee. It was like that for 2 years!!! Not trying to go too long on this story, but I was able to run 2nd half marathon last week within 2 weeks.

I started my journey back in April. Now that we are in August I’m running about 35-40 miles a week and lifting back at the gym 5 days a week with no back pain training for a full marathon… and I’m in ZERO pain thank you juvent! No more surgeries, no more shots, no more painkillers…life is great! Juvent Health everyone needs to know about this, not even kidding my health took a 180."
Evan W.
Thanks Juvent! W. A. Brennan, MD
I heard about Juvent from one of it’s developers professionally. When my 65 years started to take it’s toll in form of foot arthritis, I was desperate. A orthopedic foot and ankle specialist offered surgical fusion. That was going to require no weight bearing after surgery for 6 weeks. As a spine surgeon I would have had to put my practice on hold for 6 weeks or longer and that would have been significant damage to the practice. I spoke to the folks at Juvent, and they allowed me to purchase one for my home. About 2-3 weeks into daily use for 20 minutes each day, the foot pain started going down. After 2 months, I was no longer walking with a subtle limp. My surgery that I cancelled was three months ago. I had found something to keep me operating and walking 4000-4500 steps a day with no limp. I’ve reduced my prescription anti-inflammatory down to 1000 mg aspirin in the morning and my daily Juvent treatment. The one time I backed off it for a week, the pain came slowly creeping back. Restarting the Juvent treatments the pain subsided in about a week and things are balanced now. Thanks Juvent!
W. A. Brennan MD - Vanderbilt trained neurosurgeon
JUVENT IS A LIFE CHANGER!! - May 22, 2023

After a lifetime of downhill snow skiing, biking, swimming, water polo, hiking which continued into my early 50’s, I found myself in my mid-50s with such severe joint pain in shoulders, hips, and right knee (which had an anterior cruciate ligament reconstruction in my 20’s) that I couldn’t even walk without excruciating pain and sleep had become impossible.
After consulting with an orthopedic surgeon to see if there were any treatment options to bring some relief, I was recommended to take high dose non-steroidal anti-inflammatories until the pain is unbearable and then knee replacement. The pain medications did not bring relief and symptoms worsened.
A month after this consultation, I was tackled by two 75-pound dogs running at full speed making direct impact into my right knee. MRI revealed the ACL graft destroyed, tear of proximal and mid posterior cruciate ligaments, further injury to what little remained of the menisci, degenerative/arthritic change with chondral loss with subchondral bone marrow edema.
I was thankful I didn’t fracture my femur or hip with the force I was hit. Following this injury, I was in even more pain plus marked swelling and instability of the knee. I was given crutches to use, however, this aggravated the pain in my shoulder. Severe sleep deprivation caused by the pain left me in deep misery and severe depression.
My primary care doctor who had a family member get relief using Juvent recommended I try it to see if I might find some improvement. I can never thank him enough for this suggestion! With the post-injury swelling and pain, I was not initially able to stand on the Juvent for 20 minutes, so I started with just 5-10 minutes a couple times each day, extending the time as I could tolerate it. In less than two weeks, I was able to stand for the full 20-minute cycle without any breaks. I definitely experienced improved sleep within the first 5 days of use. I cut back in both frequency and dose of pain medication, and after about 5 weeks of using the Juvent 20 minutes/day for 6 days/week, I stopped taking all pain medications. In addition to getting relief where I was hurting the most (my knee), I was also getting relief in my hip and my shoulder. I also noticed that the chronic feet and ankle pain that had come with age had diminished. I also noticed upper and lower leg muscles were less achy.
Prior to using the Juvent, I was struggling to accept the fact that age had taken a toll on my body after a lifetime of high impact sports compounded by the recent injury. I would have to navigate as best I could to minimize standing and walking time to avoid excessive swelling and pain.
It has been three months since initiating Juvent use. I was able to walk a mile this week without pain and not “paying for it “afterward as I had prior to the Juvent.
Not only have I found tremendous benefit, but my family has also. My husband (early 60’s) experienced relief in his knees and ankles. He finished this ski season feeling better than he can remember. Our daughter who is a figure skater has used it for a tender knee and back strain. After 1-2 treatments she was completely pain free. What a gift to not take medications to cope with the pain.
There are not enough words to adequately express what a life-changer Juvent has been for me and for my family. Thank you to all who created it and make it available for me to benefit and have in my own home!
I only wish I knew about Juvent about 10 years ago to help my father with neuropathy, knee pain, and balance in his final years. I am convinced that he would have had life-changing relief and improved quality of life.
I thank God for all of you at Juvent!!! ~ Cathy C., California

USING SPACE-AGE TECHNOLOGY TO LIVE LIFE TO ITS MAXIMUM
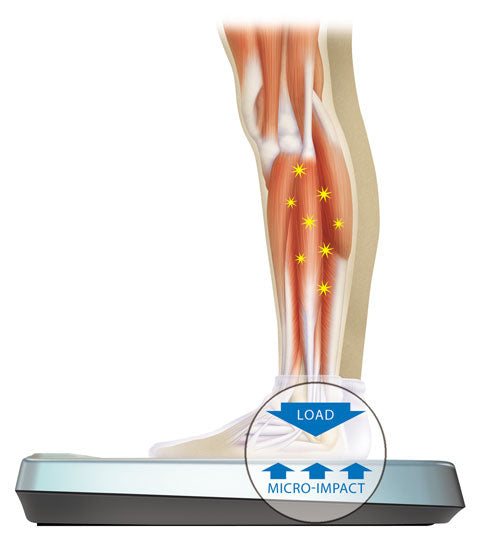
We know from our space program that it is impossible to achieve total health without a sufficient amount of skeletal impact. It’s that simple. NASA funded the early research that resulted in the Juvent Platform technology.
Your bones have many functions that rely on regular healthy impact, it’s one of the fundamental requirements for optimal health. Juvent makes it possible to get daily skeletal impact that is safe, effective and easy.
Good health is so important and the Juvent Platform can help to maintain yours. It’s evidence- based — rooted in extensive research — and it’s groundbreaking. Easy and effective, the Juvent Micro-Impact Platform is an effective solution that promotes musculoskeletal health while helping to relieve joint pain and improving balance.
Simply standing on the Juvent Platform for 10 to 20 minutes a day. You’ll feel a subtle, pleasant stimulation. Starting at your feet and legs, it extends upward through your torso, arms, and upper body. Using our platform just three days a week has been shown to help improve blood flow, ease joint, knee and back pain, and increase stability.






Juvent
Juvent Micro-Impact Platforms
Our original Juvent featuring low-magnitude mechanical stimulation (LMMS). 10-year limited warranty. See which Juvent is right for you via our comparison guide!
Perfect for athletes. The Pro HD platform features our original Juvent technology with a sleeker look and solid stainless steel top plate for greater rigidity and durability. 10-Year limited warranty. See which Juvent is right for you via our comparison guide!
Designed for physicians, physical therapists, and trainers. The clinical HD platform with ASSIST features our original Juvent technology with a solid stainless steel top and the option to turn it on with a remote, wired foot switch for those that cannot stand. It has a 10-minute cycle and resets immediately following the end of treatment. 3-Year limited warranty. See which Juvent is right for you via our comparison guide!
Our original Juvent with an aluminum Zarges Executive rolling travel case. Retractable handle and wheels. TSA approved for flights.
Share

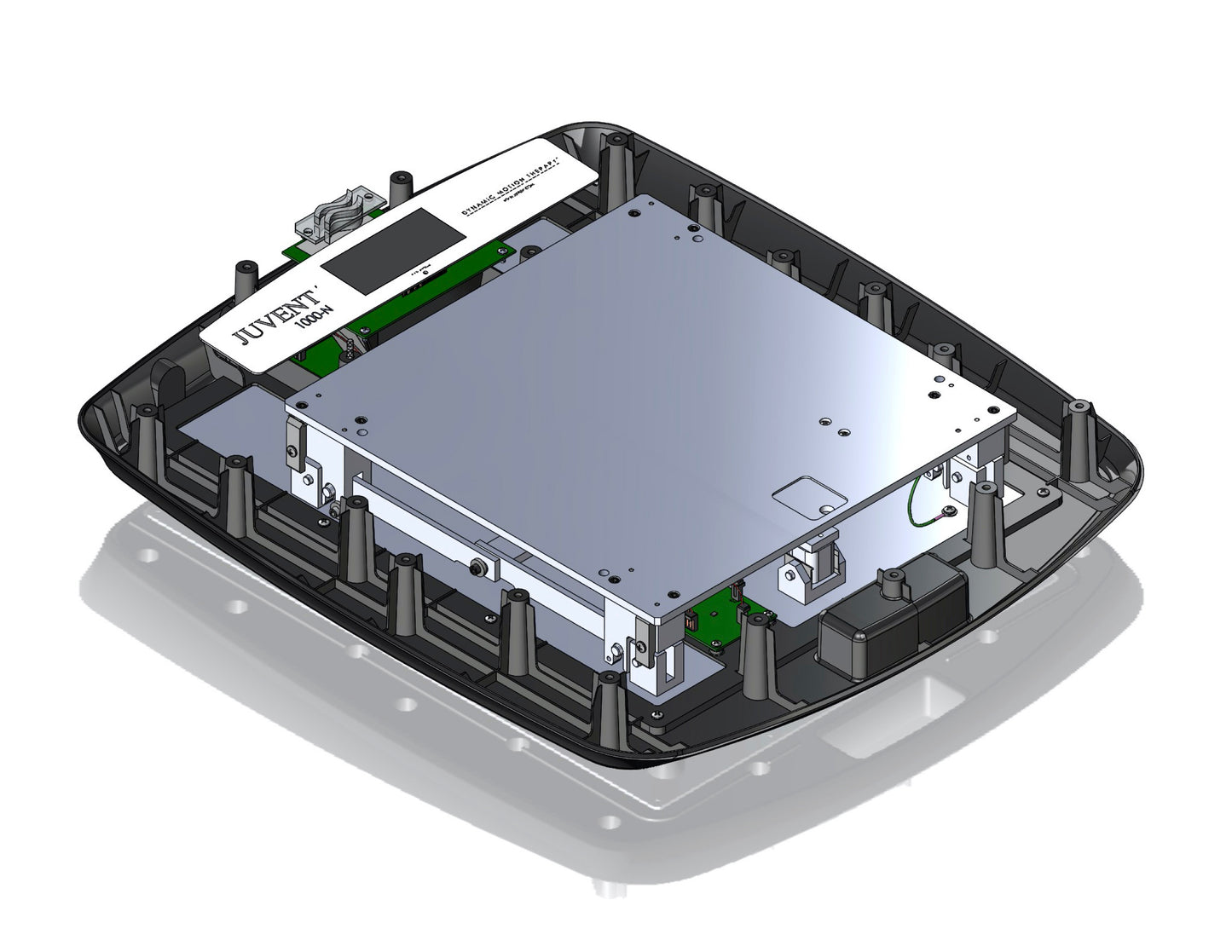
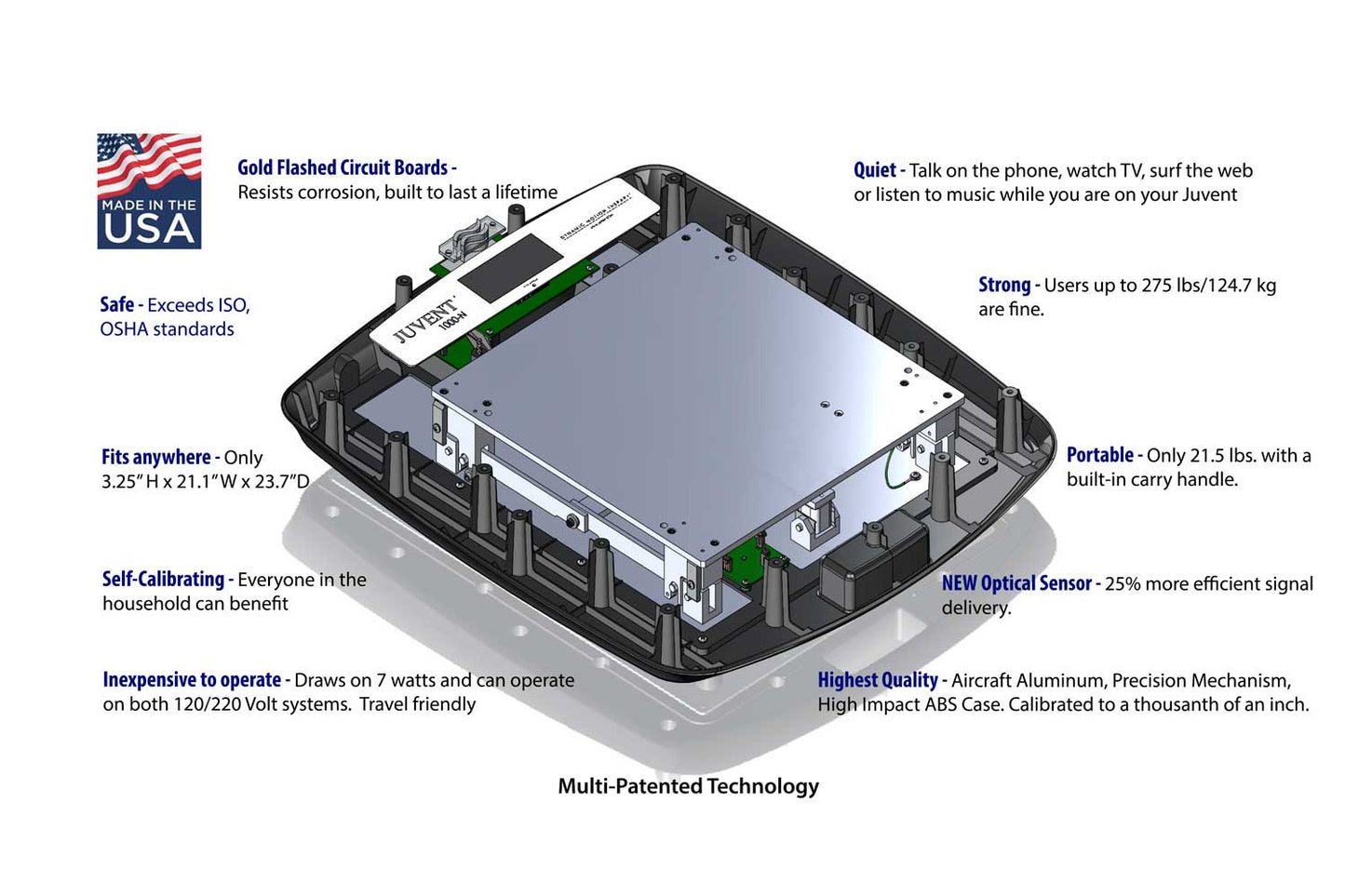




Additional Information
Unboxing Video
Technical Specifications
- The Juvent Health Micro-Impact Platform® is a precision FDA Class 1 medical exercise and rehabilitation device.
- Weighs 21.5 lbs, with a standard operating capacity of 250 lbs.
- Natural non-invasive treatment
- Quiet. Talk on the phone, watch TV, surf the web or listen to music while on Juvent.
- Everyone in the household can use the Juvent since it is self-calibrating.
- Compact for easy storage, portable and durable (Height 3.25 in / Width 21.1 in / Depth 23.7 in)
- Strong. Maximum capacity of 275 lbs /124.7 kg
- Energy-efficient – requires only 7 watts
- 120/220 Volt
- Custom Colors available
In the US it is considered investigational for the treatment of osteoporosis or improvement/maintenance of bone mineral density. JUVENT products are covered by the following issued patents, pending patents and their foreign equivalents: 6,843,776 6,884,2277,094211 7,207,954 7,207,955 7,985,191 8,114,036 11/369,611 11/486,538.
Watch a Juvent being built
Read the Owner's Manual
Featured products
-
Juvent Micro-Impact Platforms
Regular price From $5,995.00 USDRegular priceUnit price / per -
Factory Certified Pre-owned Juvent Micro-Impact Platforms
Regular price $4,995.00 USDRegular priceUnit price / per$5,995.00 USDSale price $4,995.00 USDSold out
New Products
-
Powerblock Sport 24
Regular price $199.00 USDRegular priceUnit price / per -
British Berkefeld System EXCLUSIVE BUNDLE
Regular price From $311.46 USDRegular priceUnit price / per